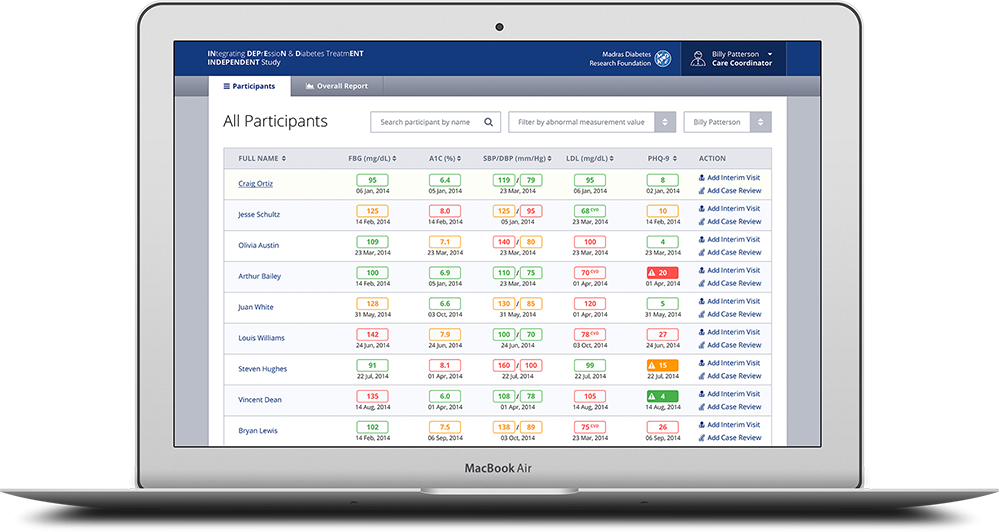
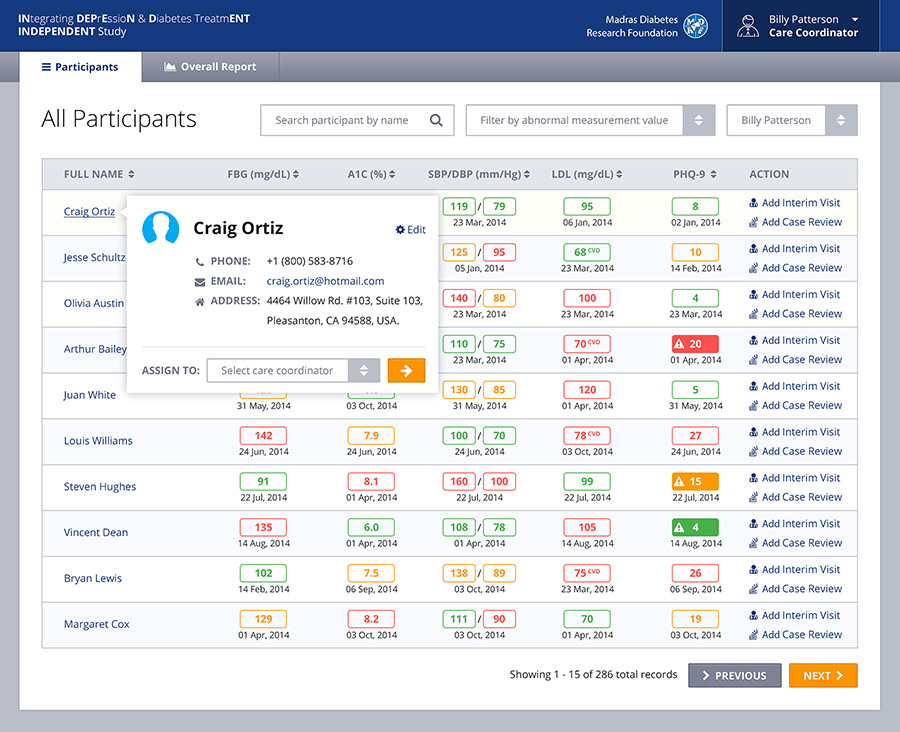
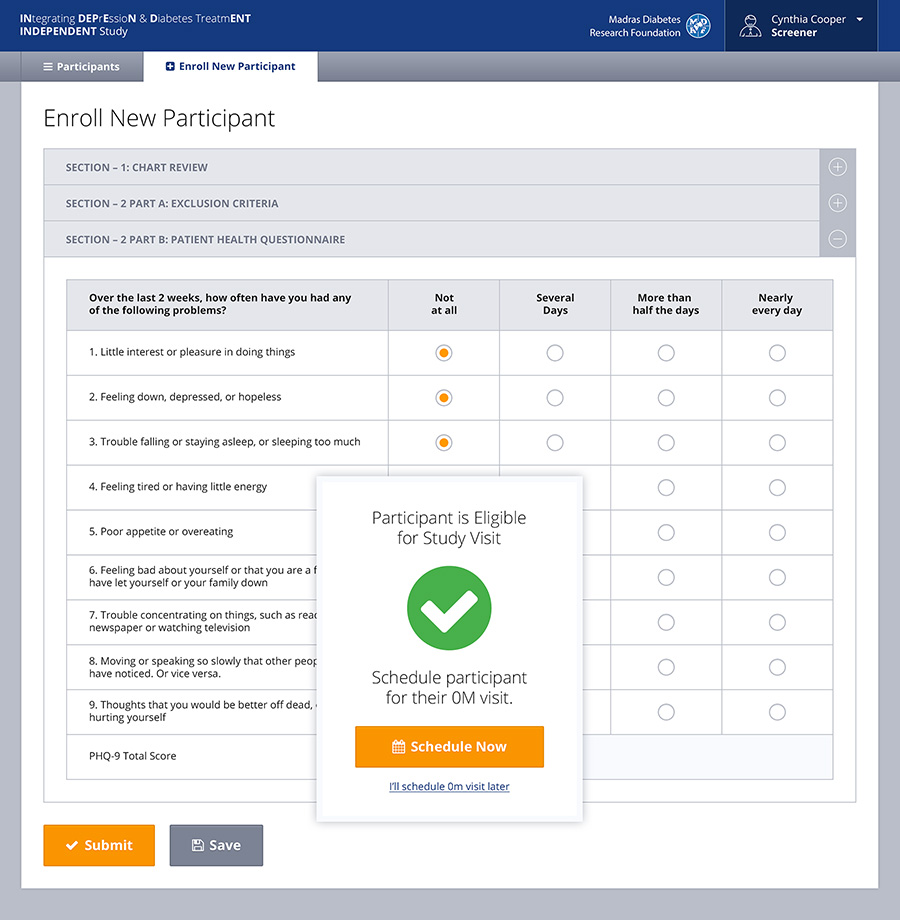
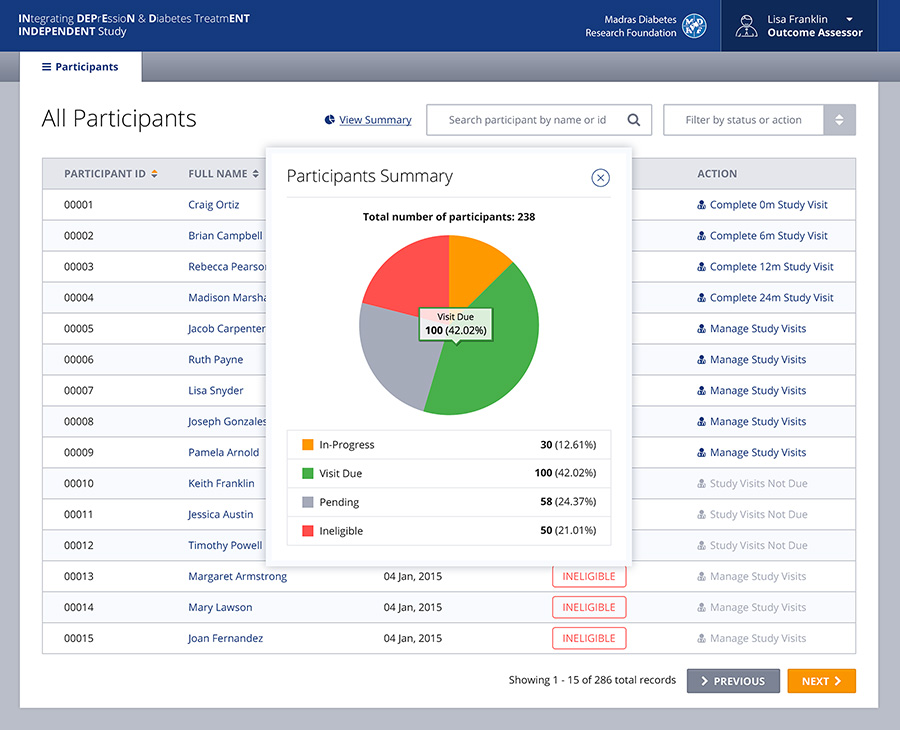
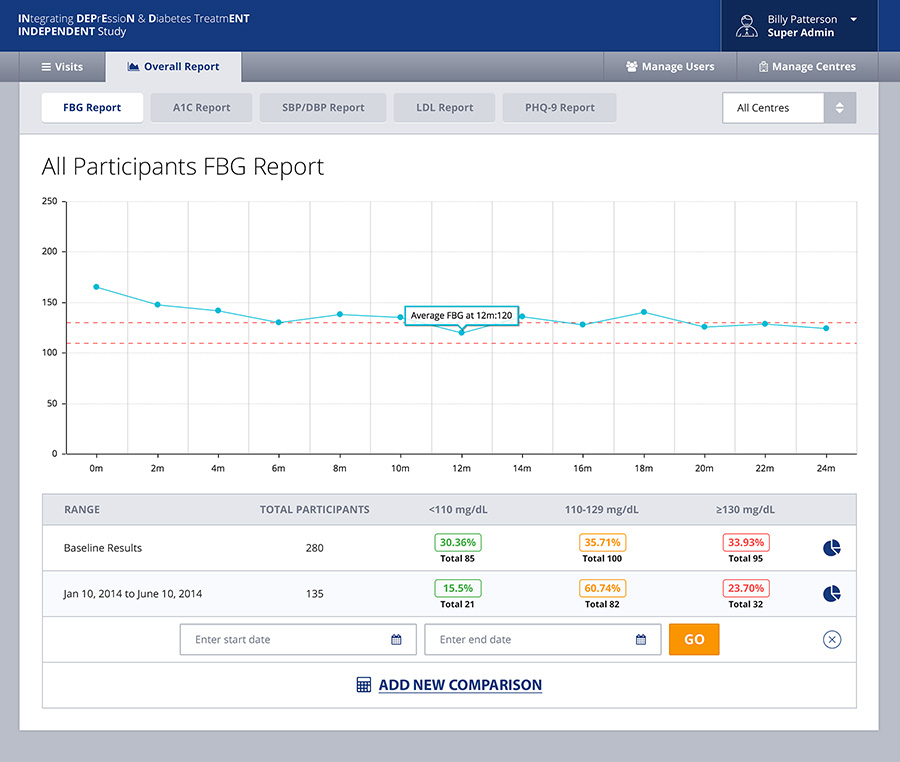
Built as a very user friendly and intuitive app for end-users who might not be the most technically savvy, the front-end system allows for caregivers, doctors, hospital admin staff or others to capture information about the patient’s condition at many different stages of the trial. The back-end of the system allows physicians to generate and view reports, review treatment prescribed and even get recommendations from the system itself based on the symptoms. This is thanks to a custom built algorithm in the back-end which required a lot of cross collaboration between Emory and Cygnis in order to build.
Design considerations were made to ensure that the application is usable on tablet computers as well, which allows doctors, nurses, physicians and others using the system to be able to do so in a mobile manner. All dashboards and other interactive elements were created keeping cross platform compatibility in mind.
 Web Applications
Build web apps using cutting-edge technology
Web Applications
Build web apps using cutting-edge technology
 Business Intelligence Apps
Empower your business with fast & actionable BI Apps
Business Intelligence Apps
Empower your business with fast & actionable BI Apps
 Mobile Applications
Build cross-platform apps for iOS and Android devices
Mobile Applications
Build cross-platform apps for iOS and Android devices
 Internet of Things Apps
Streamline your operations with cloud-based IoT apps
Internet of Things Apps
Streamline your operations with cloud-based IoT apps
 AI Products
Unlock the power of AI & ML with our expertise
AI Products
Unlock the power of AI & ML with our expertise
 Minimum Viable Product (MVP)
Mitigate risks & accelerate your project development
Minimum Viable Product (MVP)
Mitigate risks & accelerate your project development
 Enterprise Software
Build custom enterprise solutions for your business
Enterprise Software
Build custom enterprise solutions for your business
 Software as a Service (SaaS)
Scale your business with ease and cost-efficiency
Software as a Service (SaaS)
Scale your business with ease and cost-efficiency